 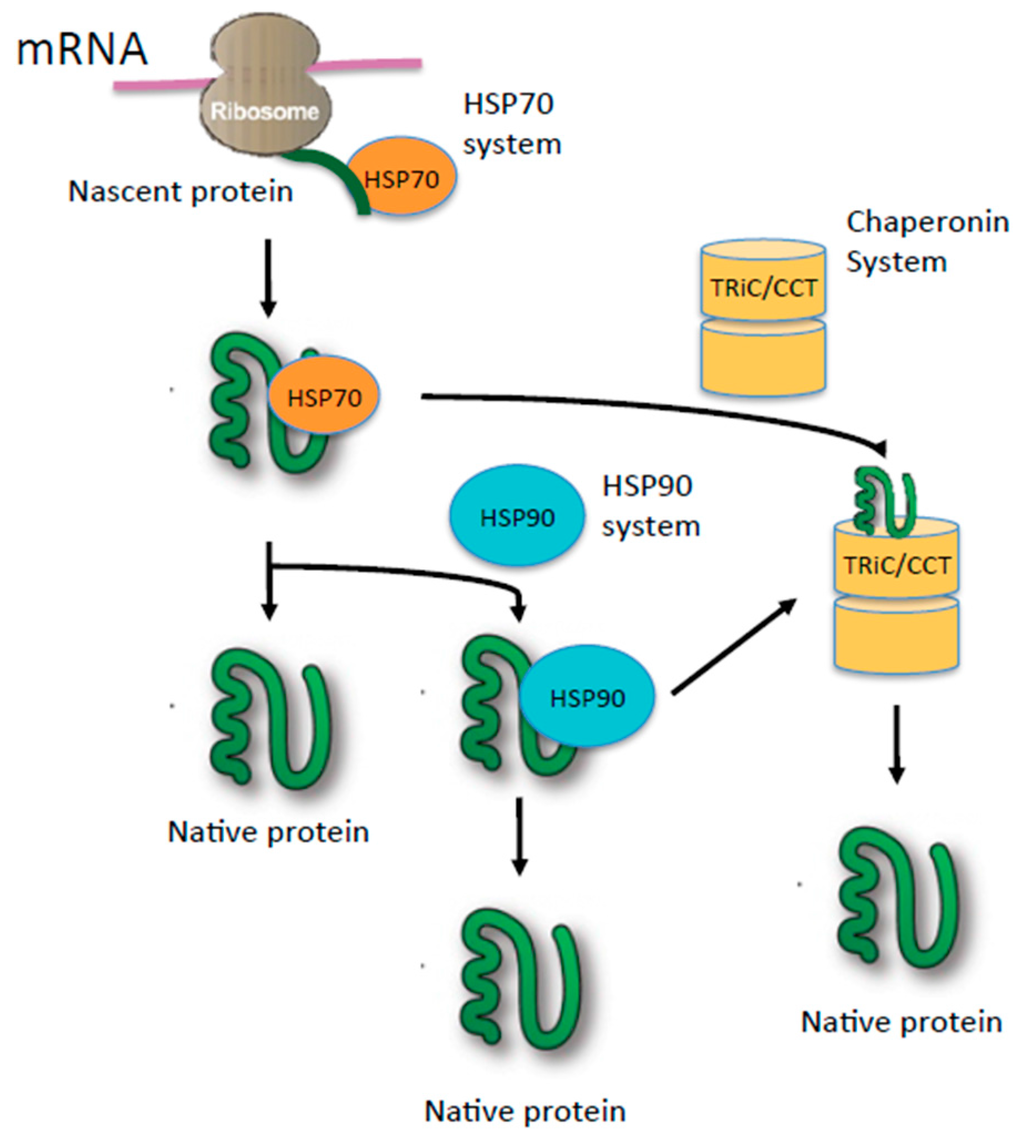
It is widely noted that the dysregulation of stress proteins is associated with a variety of human diseases, including cardiovascular diseases, neurodegenerative diseases (e.g., Parkinson’s diseases, Alzheimer disease), stroke, human cancers and infectious diseases. Responses triggered by SPs can either activate pathways to promote cell survival or initiate cell death (i.e., apoptosis, necrosis, pyroptosis or autophagic cell death) for eliminating the damaged cells to protect a particular organ/tissue under given conditions. 1 SPs are ubiquitously expressed in all kind of cells, triggering signal cascades for neutralizing and eradicating the stresses occurring both intracellularly (e.g., pathogen invasion) and extracellularly (e.g., starvation, stimulation by cytokines/chemokines or hormones). Stress proteins include heat shock proteins (HSPs), RNA chaperone protein (RNPs), and proteins mainly function in the endoplasmic reticulum (ER): peptidyl-propyl isomerases, protein disulfide isomerases (PDIs) and the lectin-binding chaperone system. They exhibit protective effects against stresses. Stress proteins (SPs) are a diverse group of proteins that are synthesized at increased levels when cells are exposed to either intracellular or extracellular stressful stimuli. As SPs also attract a great interest as potential antiviral targets (e.g., COVID-19), we also discuss the present progress and challenges in this area of HSP-based drug development, as well as with compounds already under clinical evaluation. In this review, we summarized the biologic function of stress proteins, and current progress on their mechanisms related to virus reproduction and diseases caused by virus infections. Dysregulation of stress proteins is associated with many human diseases including human cancer, cardiovascular diseases, neurodegenerative diseases (e.g., Parkinson’s diseases, Alzheimer disease), stroke and infectious diseases. hnRNPs involve in a large number of cellular processes, including chromatin remodelling, transcription regulation, RNP assembly and stabilization, RNA export, virus replication, histone-like nucleoid structuring, and even intracellular immunity. RNA chaperones are a group of heterogeneous nuclear ribonucleoproteins (hnRNPs), which are essential factors for manipulating both the functions and metabolisms of pre-mRNAs/hnRNAs transcribed by RNA polymerase II. Regarded as a double-edged sword, HSPs also cooperate with numerous viruses and cancer cells to promote their survival. The major functions of HSPs include chaperoning misfolded or unfolded polypeptides, protecting cells from toxic stress, and presenting immune and inflammatory cytokines. Stress proteins (SPs) including heat-shock proteins (HSPs), RNA chaperones, and ER associated stress proteins are molecular chaperones essential for cellular homeostasis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
